Specific latent heat (symbol $l$) is the amount of heat energy required to convert unit mass ($1\ kg$) of a substance from one phase to another. It is an intensive property. The associated formula is: $$ \begin{equation}\begin{aligned} Q=ml\\ \end{aligned}\end{equation} $$ Thus: $$ \begin{equation}\begin{aligned} l=\frac{Q}{m}\\ \end{aligned}\end{equation} $$
Example
It requires $4,600,000\ J$ of energy to convert $2\ kg$ of water to that same mass of steam. Find the specific latent heat of vaporization of water.
The SI Unit for specific latent heat is thus $Joules\ per\ kilogram\ (Jkg^{-1})$.
Types of specific latent heats
- Specific latent heat of fusion ($l_f$) is the heat energy required to convert unit mass ($1\ kg$) of a substance from its solid state to its liquid state.
- Specific latent heat of vaporization ($l_v$) is the heat energy required to convert unit mass ($1\ kg$) of a substance from its liquid state to its gaseous state.
Latent heats are associated with phase changes.
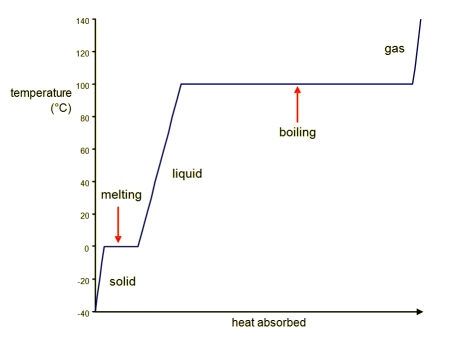
Heating Curve for water
This graph shows us the various temperature changes and phase changes associated with the heating of water.
Credits: Course Hero- When there is a temperature change ($Q=mc\Delta T$), there is no phase change ($Q=ml$) and vice versa
- Sections of the graph which are horizontal (gradient is zero) represent phase changes
- Sections with non-zero gradients represent temperature changes