Because specific heat capacity and specific latent heat are intensive properties, it is important that we have reliable means of measuring their values.
Determining the specific heat capacity
There are two common ways of finding the SHC of a substance:
- Electrical method
- Method of mixtures
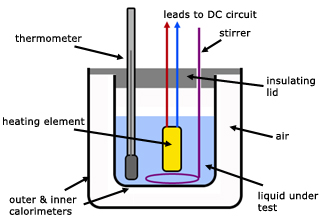
Electrical method
- Suitable for liquids
- Measure the mass $m$ of the substance
- Measure the initial temperature $T_i$ of the substance
- Immerse a power heater (immersion heater) with voltage rating $V$ and current rating $I$ into the substance
- Heat the substance for an amount of time $t$
- Measure the final temperature $T_f$ of the substance
- The heat energy supplied to the substance will be $$ \begin{equation}\begin{aligned} E_{heat}=VIt\\ \end{aligned}\end{equation} $$
- Use the formula $E_{heat}=mc(T_f-T_i)$ to find the specific heat capacity $c$ of the substance
- The specific heat capacity will thus be: $$ \begin{equation}\begin{aligned} c=\frac{VIt}{m(T_f-T_i)}\\ \end{aligned}\end{equation} $$
Method of mixtures
Video:
Loading video
- Suitable for solids especially metals
- Measure the mass $m_s$ of the solid
- Measure the mass $m_w$ of the water in styrofoam cup
- Place the solid in boiling water for a few minutes to allow the initial temperature of the solid to reach $100\degree C$
- Measure the initial temperature $T_{room}$ of the water in the styrofoam cup
- Transfer the solid from the boiling water to the water in the styrofoam cup
- Keep track of the temperature of this mixture until the water and solid reach a final mutual temperature $T_f$
- The heat energy gained by the water will be
- The heat energy gained by the solid will be
- The heat energy lost by the solid will be the negative of the heat energy gained by the water:
- Thus the SHC of the solid will be